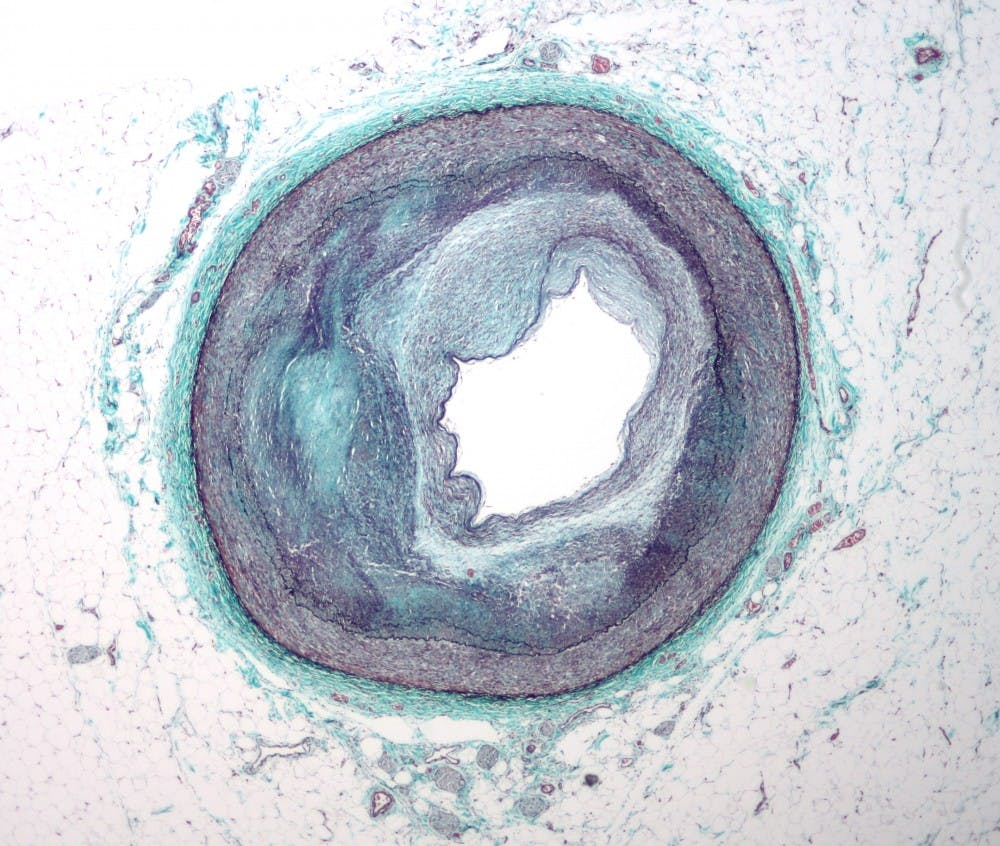
After more than a year of research and control analysis, researchers at the Medical School had a paper published January in the research journal Nature Methods. Their research — which has the potential to help doctors better understand artherosclerosis — describes a new way to identify distinctive characteristics of DNA modification in the smooth muscle cells of mice by combining two preexisting procedures.
The laboratory members working under Dr. Gary Owens, professor of molecular biological physics, examined epigenetic mechanisms in smooth muscle cells — the alteration in specific gene locations of proteins that help package strands of DNA for gene expression. The researchers also devised new methods to visualize what differentiates one individual cell from another.
Epigenetics, or the study of gene expression and cellular phenotype, provides ways of detailing how cells become varied and specialized within an organism. “Every cell contains the same DNA, [but] what makes a brain cell different from a muscle cell is epigenetics,” said Ahn T. Nguyen, researcher on the project and post-doctoral fellow.
For their epigenetic research, the University researchers focused on histones, proteins that add structure to DNA molecules. Unique patterns of histone modification are present within each type of cell. The goal for researchers and doctors is to be able to distinguish cell type based on epigenetic marks.
Nguyen said this new research looks at a smaller-than-normal sample size to better understand why some cells behave differently than others. Researchers labeled “good” cells, which were functioning properly, to distinguish them from “bad” cells in the mice.
Since good cells behave normally, the team was able to identify them and study how their histones acted compared to the histones in the bad cells. “Previously, the only way to identify histone modification was with at least 1,000 cells,” Nguyen said. “[Our] method looks at histone modification in only one cell … [This] helps us see which individual cells have [the labels].”
Before the University researchers developed their new method of studying protein and DNA interaction, scientists used a process called chromatin immunoprecipitation in which researchers bonded the DNA and protein together, cut the bonded sections off and extracted specific DNA sequences from the combination. ChIP requires sorting through an epigenetic analysis of thousands of cells, but the University researchers’ improved method uses fewer cells and yields more precise results to more easily identify cell type. This new method combines labeling DNA sequences and detecting protein interactions to allow researchers to look at the process regulating the expression of genes in individual cells.
The applications of the new recognition technique are expansive. Because it serves as a more efficient and clear method to analyze an individual cell’s epigenetic markers, the researchers predict that the technique will help scientists develop their understanding of atherosclerosis — a common disorder that occurs when fat and cholesterol build up in arteries and block blood flow — by allowing them to pinpoint smooth muscle cells’ role in the process.