Researchers of the Department of Surgery at Uva School of Medicine have received five grants that aim to increase the number of lungs for transplant and to prevent and treat complications that arise after lung transplantation. Funded by the NIH, the research focuses on developing new ways to recondition injured lungs that once could not have been used for successful transplants.
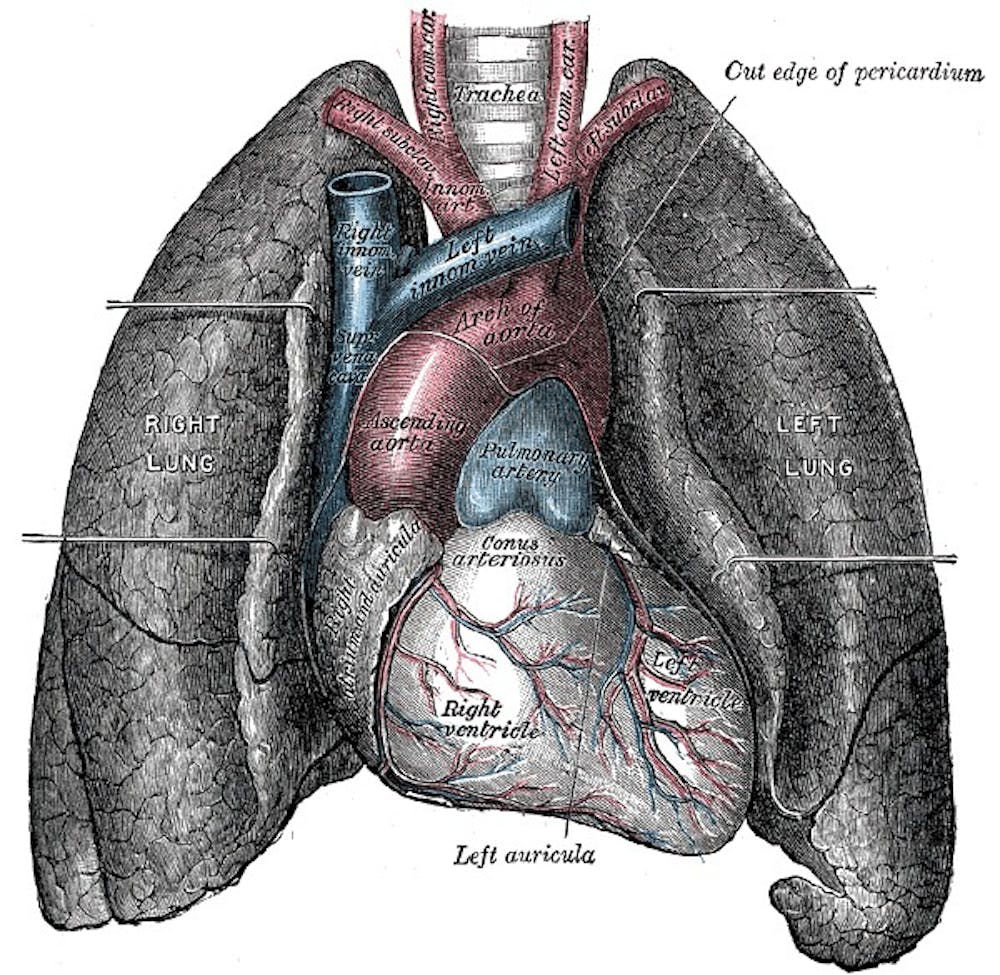
In lung transplantation, a patient’s diseased lungs are partially or completely removed and replaced with one from a donor. Transplantation is usually
Lung transplant surgery has its limitations. Donor lungs usually come from patients who die by brain death, as their lungs are still viable and functioning. This excludes populations of patients who die by other means, such as heart attacks.
“There are many patients that undergo a heart attack outside or even within the hospital that, if they arrive and they’re dead, the surgeons will not use their lungs for
Rejection and infection are serious complications that arise from transplantation and limit the effectiveness of the surgery. According to the Lung Institute, while up to 78 percent of transplant patients survive their first year, only about 50 percent survive five years.
One of the problems that
However, Laubach, University of Virginia’s Dr. Irving Kron and their colleagues may be able to increase the number of
"This research started from an early transplant I performed at UVA around 1990,” Kron said in an email statement. “A young patient had an an uncomplicated transplant but nearly died because the lung didn't work temporarily.”
One aspect of the research aims to increase viable lungs for transplant using a pig model. Researchers induce a heart attack in pigs to ensure that the injury affected the lungs and intervene by using an ex vivo perfusion system. This system has a pump that injects special buffer solution into the lungs and also ventilates them in order to get oxygen into the once oxygen-deprived lung tissue. Often, after the lungs are hooked up to the system for several hours, the lungs acquire sufficient oxygenation and may be used in transplant patients.
Not all lungs for transplant get better with this system. Lungs with too much inflammation are unsafe to be used for transplant even after they are on the ex vivo perfusion system.
Laubach and Dr. Lau, a cardiothoracic surgeon at the University, are using a therapeutic approach with drug therapy to help alleviate acute and chronic inflammation. Their research involves a clinical trial that tests a drug called regadenoson, which can target adenosine-2A receptors on the lungs.
These receptors bind to adenosine, which is released by cells when they are inflamed. Regadenoson is a potent anti-inflammatory molecule that limits lung inflammation even when adenosine is released.
Another one of the grants obtained by Laubach and Dr. Kron funds their research into improving detection of IR injury to increase the survivability of the lungs in transplant patients.
Laubach and Kron hypothesize that targeting cells early in the inflammatory process, such as neutrophils and macrophages, can help in early detection of acute and chronic IR injury so that the injury can be treated quickly.
Laubach and Kron’s research can be applied to other fields other than pulmonology.
“The imaging techniques, if they are successful in our lab,
The fourth grant, whose
The fifth grant, headed by Laubach, targets
“That is a very basic project that if it pans out could lead to another target on how to prevent injury by developing molecules that prevent
Obtaining this grant not only required years of research but also researchers willing to cooperate on their discoveries.
“We are a collaborative group—a team—of researchers and clinicians in a department,” Laubach said. “ We all have common goals of helping these transplant patients but we are going about it in different ways.”