The process of neurodevelopment is responsible for creating neurological pathways in the brain that are responsible for controlling our movements, body functions and helping us retain memories. According to the Brighton Center for Pediatric Neurodevelopment, neurodevelopment sets a person up for the rest of their life and is a critical function. During this process, over half of the neurons die, a process called cell cleanup or dieback, in order to ensure the strongest neurological connections. A group of University neuroscientists and researchers discovered the importance of the AIM2 cell cleanup pathway as it relates to faulty cell cleanup and possible reactivation later in life.
Dr. John Lukens, assistant professor of medicine and principal investigator, notes the importance of neurodevelopment.
“If there's any kind of perturbations or changes in the kinetics, it can have long lasting issues that lead to things like neurodevelopmental disorders like autism and schizophrenia,” Lukens said.
There are a variety of issues that lead to too much or little cell cleanup. According to Lukens, early infections soon after birth, exposure to neurotoxic chemicals, oxygen levels in the brain and other infections such as Zika virus can all have negative impacts on the development.
“If you have a neuron that has issues straight out of the gate, it has all these errors in the DNA that's basically going to have a result in a faulty neuronal connection being in the brain … from a very early time point,” Lukens said.
The John Lukens Lab, where the research project took place, is part of the neuroscience department at the University and focuses on immunological pathways and the development of neurodegenerative, neurodevelopmental, mental and behavioral disorders.
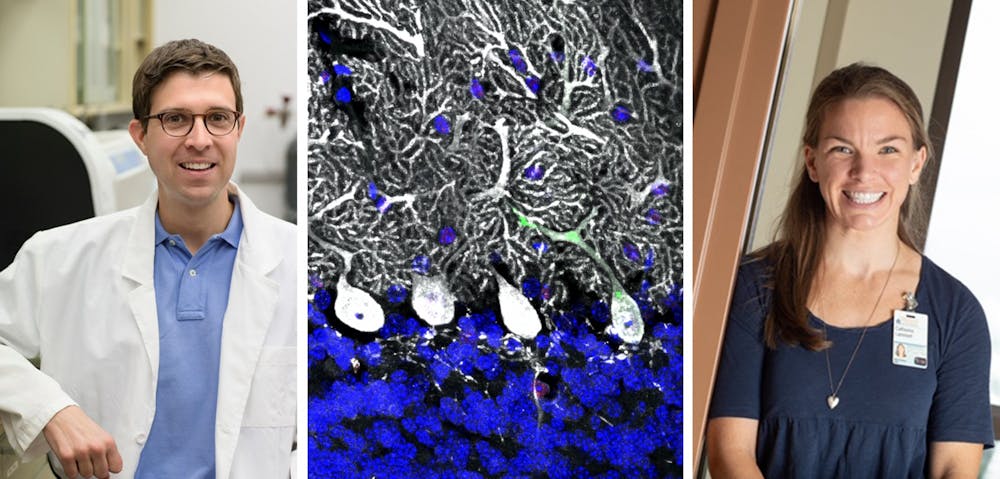
The research team — consisting of neuroscience graduate student Catherine Lammert, postdoctoral fellow Elizabeth Frost, undergraduate lab researcher Calli Bellinger, neuroscience graduate student Ashley Bolte, Washington University neuroscience graduate student Celia McKee, data science graduate student Mariah Hurt, cognitive sciences undergraduate student Matt Paysour, neuroscience graduate student Hannah Ennerfelt and Lukens — discovered a pathway that helps to control DNA damage and ensure proper neurodevelopment. Their discovery was somewhat of a chance discovery stemming from unexpected results from their initial focus on traumatic brain injury.
Before moving forward with the initial trauma experiment, they tested mice to measure their motor coordination and discovered that mice and other animals lacking a specific cell cleanup pathway, AIM2 inflammasome, performed very poorly. The molecular pathway detects neurons that are faulty in order to ensure dangerous neurons do not get incorporated to the brain.
The team found that a lot of people with mutations or those that lack the AIM2 inflammasome have neurodevelopmental disorders amongst other diseases.
“Manipulating this pathway to help the developing nervous system to eliminate those cells could potentially provide at least biomarkers or therapeutic targets to treat neurodevelopmental disorders like autism,” Lukens said.
The use of the pathway could potentially be useful later in life as well as earlier in life.
As people grow older, the pathways that repair DNA become less effective. According to Lukens, it is thought that the acquisition of DNA damage overtime causes the degenerating of our organs, or atrophy.
As there are currently no treatments that treat the underlying causes of neurological diseases like amyotrophic lateral sclerosis or Alzheimer’s, the possibility of reactivating the pathway later in life could potentially hold back neurodegeneration.
Lukens also emphasized the pathway can get inadvertently reactivated in old age due to DNA damage and if not controlled, can have negative consequences.
Lammert, who worked on the project for just under five years, thinks that treatments are far away but notes that many papers have shown inflammasomes to be important.
“I think that the main really cool thing about this finding is that inflammasomes, which usually are activated in response to an infection or some kind of exogenous danger signal actually respond in the brain just at baseline and not responding to any disease or damage,” Lammert said. “In terms of treatment for things, it's just maybe shedding light on a new pathway that could be targeted.”
The team’s discovery is the tip of the iceberg in regard to possible neurological disease treatment and prevention. The pathway could be a possible target to combat neurological diseases such as ALS or alzheimers.